Our series of Best-in-Class webinars provides a quick study on various topics related to core food safety concepts. Each course is performed as a 2-hour “lunch and learn”-style presentation. Lead instructor, Mike Cramer of the Eurofins Advanced Learning Institute, delivers practical guidance and basic tools through the viewpoint of his 40+ years of experience in meat and food manufacturing.
These free webinar sessions are valuable for food industry personnel involved in all phases of food manufacturing, distribution, retail, and storage, and are critical to the foundation of a strong, well-rounded food safety culture.
MEET THE HOST
Michael Cramer | Trainer, Eurofins Advanced Learning Institute
Michael Cramer started his career at Swift & Company in Pennsylvania as a summer employee. He worked his way up to QA Manager (Pennsylvania and North Carolina), Production Specialist and Corporate Documentation Manager (Illinois) to QA Manager at the Swift-Eckrich plant in Chicago and then QA Director for the Armour processed meat and poultry plants. He then moved to Specialty Brands, Inc./Windsor Foods/Ajinomoto Foods North America where he stayed from 1993 to his retirement in 2023. There he served as Director of Food Safety and Quality assurance before being promoted to Senior Director of FSQA.
Mike is known for his in-depth knowledge of food plant sanitation and has written a book, now in its 3rd edition, “Food Plant Sanitation, Design, Maintenance and Good Manufacturing Practices.” He is also a member of the editorial board of Food Safety Magazine and has over twenty articles published dealing with Listeria control, Biosecurity, Sanitation and Sanitary Design, Sanitation 5S, and Allergens. As well, he is a contributing member of the American Frozen Foods Institute Listeria Working Group. You can find multiple Food Safety Matters podcasts and food sanitation webinars that he has led.
Upcoming Live Webinars
Sign up for one of our upcoming live sessions. Calendar already booked up during this time? No problem! Register today, and you will receive a copy of the webinar recording within three business days after the live event.
Integrated Pest Management | June 3, 2026 | Register
Recall Policy and Action Plan | June 24, 2026 | Register
Root Cause Analysis | July 15, 2026 | Coming Soon
Regulatory Response | August 5, 2026 | Coming Soon
Listeria Management | August 26, 2026 | Coming Soon
Sign up here to be notified when registration for future episodes becomes available.
On-Demand Webinars
Check out past webinars in the Best in Class webinar series below.
In today’s supplement marketplace, contract manufacturers play a pivotal role. As retailer expectations evolve and third‑party verification becomes increasingly important, product certification is no longer just a valued add, it’s becoming a strategic advantage. Click to learn more about the benefits of product certifications.
As we look back on 2025, one thing is clear: innovation didn’t slow down, it got sharper. Across major food, beverage, supplement, and ingredient tradeshows this year, the conversation shifted away from flashy novelty and toward proof, performance, and practicality. Here is our perspective on what to expect in 2026.
How do you know when it makes sense to bring in product development support and when it doesn’t? Based on what we see across brands, ingredient companies, and startups, here’s a practical way to think about it.
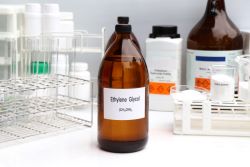
Ethylene glycol (EG) and diethylene glycol (DEG) are toxic industrial chemicals that have no place in consumer health products. Unfortunately, products ranging from cough syrups to dietary supplements have been found to contain these contaminants, often with tragic consequences.
As the category of sports nutrition matures, so does the science behind it, and few ingredients illustrate that shift better than creatine. Once viewed as a niche performance supplement, creatine has entered the mainstream, driven by a growing body of research, broader consumer education, and innovation in product formats.
While advanced technology often takes center stage in discussions about scientific accuracy, the age and preparation of samples are equally critical. By prioritizing the seemingly small details, researchers can ensure that their findings stand up to scrutiny and contribute meaningfully to scientific progress.
In the dietary supplement and ingredient industry, protecting consumer health and meeting regulatory expectations are nonnegotiable. One critical aspect of quality control is residual solvent testing. Residual solvents, volatile chemicals used during manufacturing, can remain in products if not properly removed.
When evaluating fiber testing approaches, it’s important to review how results differ across sample matrices tested by both the new and previous methods. More laboratories are implementing the Rapid Integrated Dietary Fiber Methods - AOAC Official Methods 2017.16 and 2022.01. The AOAC 2017.16 method measures the insoluble fiber fraction (IDF) and soluble fiber fraction that precipitates in 78% ethanol (SDFP) together, and separately measures the soluble dietary fiber not precipitated in 78% ethanol (SDFS) by HPLC.
Postbiotics, defined as non-viable microbial cells, their components, or metabolites that confer health benefits to the host, represent a rapidly expanding frontier in microbiome research and functional ingredient development. Watch this webinar for a comprehensive look at the rapidly evolving field of postbiotics, combining cutting-edge science, global regulatory insights, and practical testing methodologies. Originally Aired on December 11, 2025.
In this blog, Dan Brouman traces MAHA’s path since December 2024, when the MAHA Caucus launched in Congress. Brouman also discusses MAHA’s future and its effects on food policy.