Webinars
Search >>
Upcoming Webinars:
Register to attend one of our live broadcasts. Calendar already booked up during that time? No problem. Register today, and you will receive a copy of the webinar recording after the live event.
From Turnkey to Tailored: Smart Options for Managing Your IR Program
Hosted By: Anthony Lai | Vice President of Product and Market Development | Eurofins QTA Inc.
Thursday, April 16, 2026 | 12 PM - 1 PM CDT
Best in Class: Food Allergen Management
Hosted By: Michael Cramer | Trainer | Eurofins Advanced Learning Institute
Wednesday, April 15, 2026 | 11 AM - 1 PM CDT
Best in Class: Foreign Material Prevention
Hosted By: Michael Cramer | Trainer | Eurofins Advanced Learning Institute
Wednesday, May 6, 2026 | 11 AM - 1 PM CDT
On-Demand Webinars:
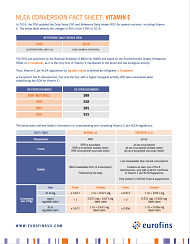
In 2016, the FDA updated the Daily Value (DV) and Reference Daily Intake (RDI) for several nutrients, including Vitamin E. The RDIs are published by the National Academy of Medicine (NAM) and based on the Recommended Dietary Allowances (RDA) of α-Tocopherol, as it is the only form of Vitamin E maintained in the blood and has biological activity. Thus, Vitamin E per NLEA regulations (or mg label claim) is defined as milligrams α-Tocopherol. Review this fact sheet for more information about reporting Vitamin E per the regulations.
The dietary supplement industry has become an increasingly regulated industry since the introduction of the dietary supplement GMPs in 2007. There is still to this day an industry confusion as to the requirements needed to comply with the dietary supplement GMP requirement to use a scientifically valid method. So does this mean the method must be validated?